Strategic Acquisition to Expand its BPH Portfolio into In-office Treatment

Olympus has acquired Israeli company Medi-tate, whose flagship product is the iTind device for the non-surgical treatment of Benign Prostatic Hyperplasia (BPH), also known as enlarged prostate.
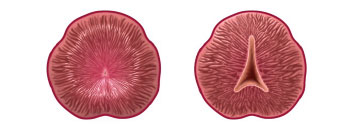
Before and after graphics of the reshaped prostate following treatment with the iTind device. The flexible three-strut nitinol device, which can be placed during an in-office procedure, gently expands over five to seven days to reshape the prostate by creating channels through which urine can flow.
Tokyo/Hamburg/Center Valley (May 28, 2021) – Olympus Corporation announced today that it has finalized the acquisition of Israeli medical device company Medi-Tate Ltd. following the announcement of its decision to exercise the call option in February 2021. Through this acquisition, Olympus expands its business line in offering in-office treatment for benign prostatic hyperplasia (BPH) treatment and solidifies its position as a leader in the field of urological devices.
Medi-Tate is a medical device manufacturer engaged in the research and development, production, and sale of devices for the minimally invasive treatment of BPH. The company’s flagship product iTind has received U.S. FDA de Novo authorization and a European CE mark. With its initial Medi-Tate investment, Olympus has had the right of distribution since November 2018.
The incidence of BPH, one of the most common diseases in aging men and the most common cause of lower urinary tract symptoms (LUTS), is expected to increase as the general global population gets older. According to the American Urological Association, BPH is a condition that nearly 80% of men will face in their lifetime, and that ratio is higher in men of older ages.ⅰ Medication and surgical treatment have been among the usual treatment options for BPH, and in recent years, more minimally invasive surgical treatment devices have come to the market.
Olympus is a market leader providing devices for BPH treatment such as resectoscopes and a wide variety of electrodes. By adding the non-ablative BPH treatment device iTind to its portfolio, Olympus can provide Urologists more treatment options according to the symptoms and needs of their patients, in this case a non-ablative, in-office day treatment that allows patients to maintain their sexual function.
Nacho Abia, Chief Operating Officer of Olympus Corporation, said, “the acquisition of Medi-Tate aligns with our corporate strategy of focusing on three priority therapeutic areas within our Therapeutic Solutions Division — gastroenterological endotherapy devices, urological devices and respiratory endotherapy devices. Medi-Tate’s innovative products offer a truly minimally invasive treatment option for patients and flexibility in the delivery of care for healthcare professionals.”
Ido Kilemnik, Chief Executive Officer of Medi-Tate, commented, “We believe Olympus appreciates our focus on long-term clinical results and the dedication of the entire Medi-Tate team. We are pleased to be joining Olympus, which shares our vision of making iTind the global standard for BPH treatment. We are excited to be collaborating with Olympus in our effort to enhance patients’ quality of life.”
Minimally invasive treatment enabled by iTind
iTind, a temporarily implanted nitinol device, supports the relief of LUTS due to BPH. The iTind treatment is performed by a urologist in either an outpatient hospital, ambulatory surgery center or in a physician’s office, where the iTind device is placed in the prostate in a folded configuration. The device slowly expands and exerts gentle pressure at three precise points to reshape the prostatic urethra and bladder neck. After five to seven days, the device is completely removed, leaving a wider opening through which urine can flow for the relief of BPH symptoms.
# # #
About Medi-Tate
Medi-Tate is an Israeli medical device company dedicated to improving men’s healthcare and quality of life via effective, non-surgical solutions for Benign Prostatic Hyperplasia (BPH). The flagship iTind treatment for BPH currently has the EU CE Mark and is approved for sale in the European Union, UK, Israel, Australia, Brazil, and is FDA cleared for use in the United States. For more information, visit www.medi-tate.com.
About Olympus’ Therapeutic Solutions business
In its Therapeutic Solutions business, Olympus uses innovative capabilities in medical technology, therapeutic intervention, and precision manufacturing to help healthcare professionals deliver diagnostic, therapeutic, and minimally invasive procedures to improve clinical outcomes, reduce overall costs, and enhance the quality of life for patients and their safety. Starting with its early contributions to the development of the polypectomy snare, Olympus’ Therapeutic Solutions portfolio has grown to include an array of surgical energy devices and a wide range of instruments to help prevent, detect, and treat disease. For more information, visit www.olympus-global.com.
i American Urological Association, Benign Prostatic Hyperplasia. Available at:
https://www.auanet.org/education/auauniversity/medical-student-education/medical-student-curriculum/bph

